
Chemistry. D and F Block - 3 Session Session Objectives 1.f-Block elements 2.Introduction to lanthanides 3.Oxidation state 4.lanthanide contraction 5.Chemical. - ppt download
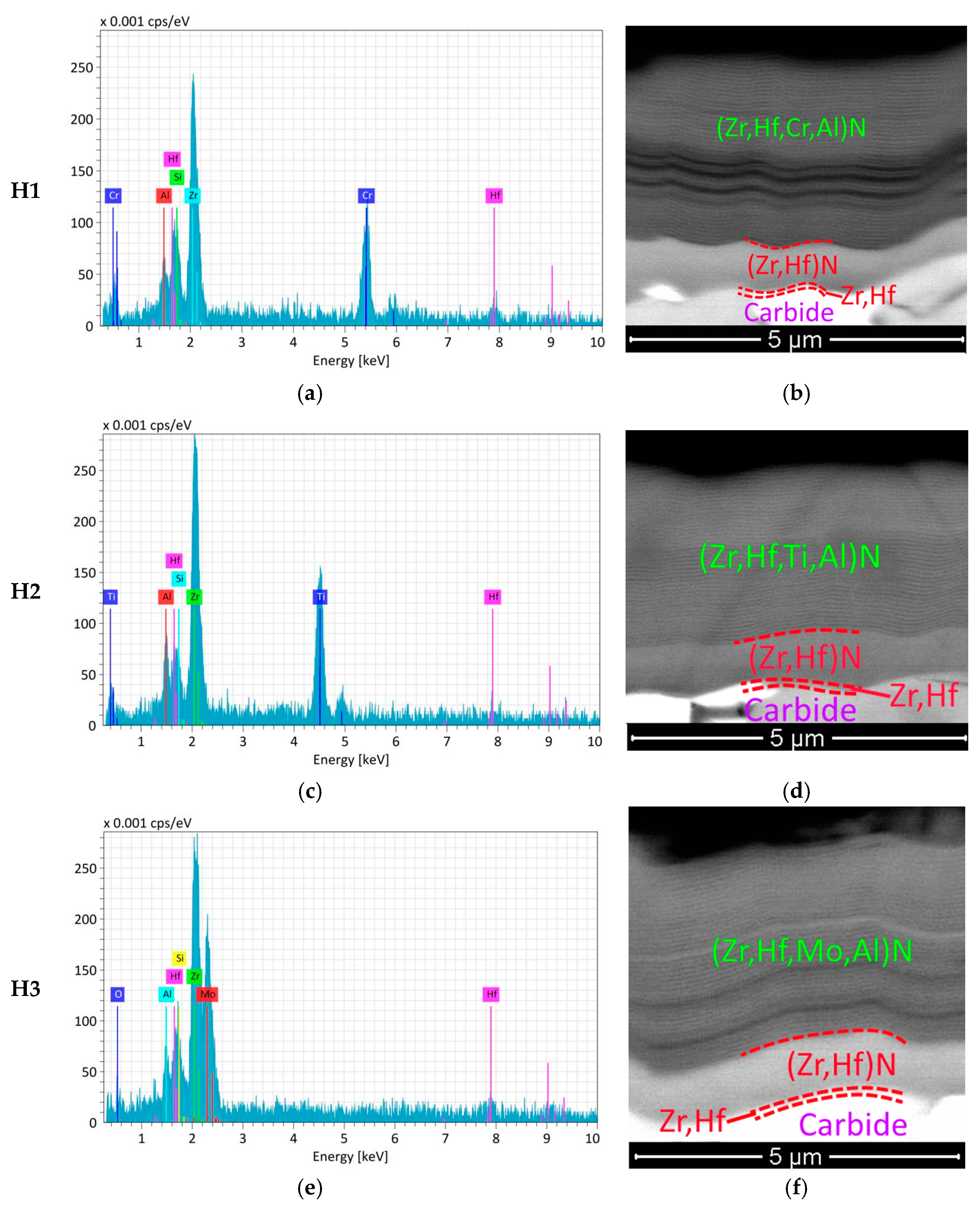
Coatings | Free Full-Text | Investigation of Properties of the Zr,Hf-(Zr,Hf )N-(Zr,Hf,Me,Al)N Coatings, Where Me Means Cr, Ti, or Mo

29. Identify a pair that has similar atomic radii. (1) Ti and HF (2) Zr and Hf (3) Cu and Hg (4) Sc and Ni

29. Identify a pair that has similar atomic radii. (1) Ti and HF (2) Zr and Hf (3) Cu and Hg (4) Sc and Ni

Comparison of the atomic radii and electronegativity of the elements... | Download Scientific Diagram

Due to lanthanide contraction Fe, Co, Ni have equal size Zr and Hf have equal size C All f-block ions have equal size D Zn and Zn+2 have almost equal size



![Bengali] Compare atomic radii of Zr (Z = 40) and Hf (Z = 72). Bengali] Compare atomic radii of Zr (Z = 40) and Hf (Z = 72).](https://static.doubtnut.com/ss/web-overlay-thumb/3591037.webp)





![Kannada] Zr and Hf have almost identical atomic radii. Give reason? Kannada] Zr and Hf have almost identical atomic radii. Give reason?](https://d10lpgp6xz60nq.cloudfront.net/ss/web-overlay-thumb/6454334.webp)
![Kannada] Zr and Hf have almost identical atomic radii. Give reason. Kannada] Zr and Hf have almost identical atomic radii. Give reason.](https://static.doubtnut.com/ss/web-overlay-thumb/5540907.webp)






