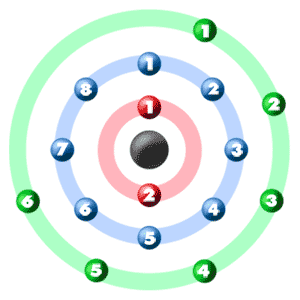
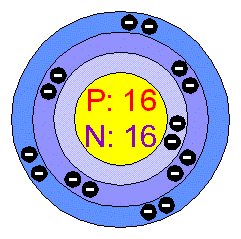
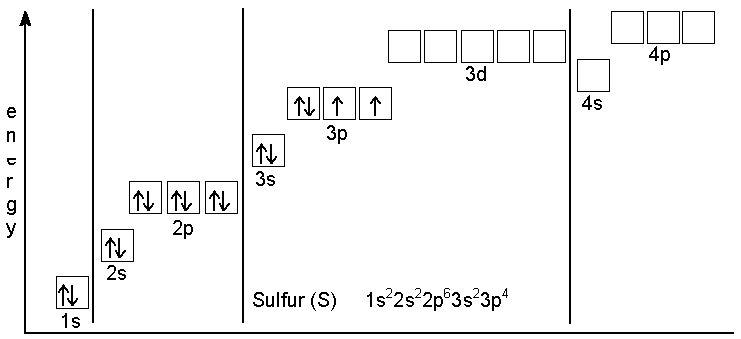
Consider a neutral atom of sulfur. There are three shells (primary quantum levels) in this atom. How many electrons does it have in: a. total b. n = 1 shell c. n =
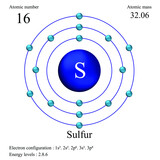
Sulfur atomic structure has atomic number, atomic mass, electron configuration and energy levels. Stock Vector | Adobe Stock
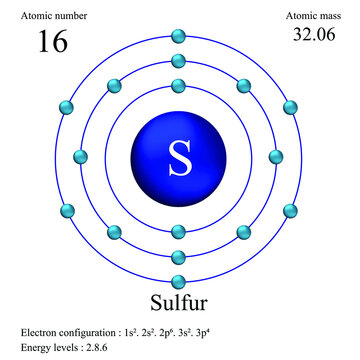
Sulfur atomic structure has atomic number, atomic mass, electron configuration and energy levels. Stock Vector | Adobe Stock
Oxygen reacts with fluorine to form only OF_2, but sulphur which is in the same Group 16 as oxygen, reacts with fluorine to form SF_2, SF_4 and SF_6. Why is this? | Socratic
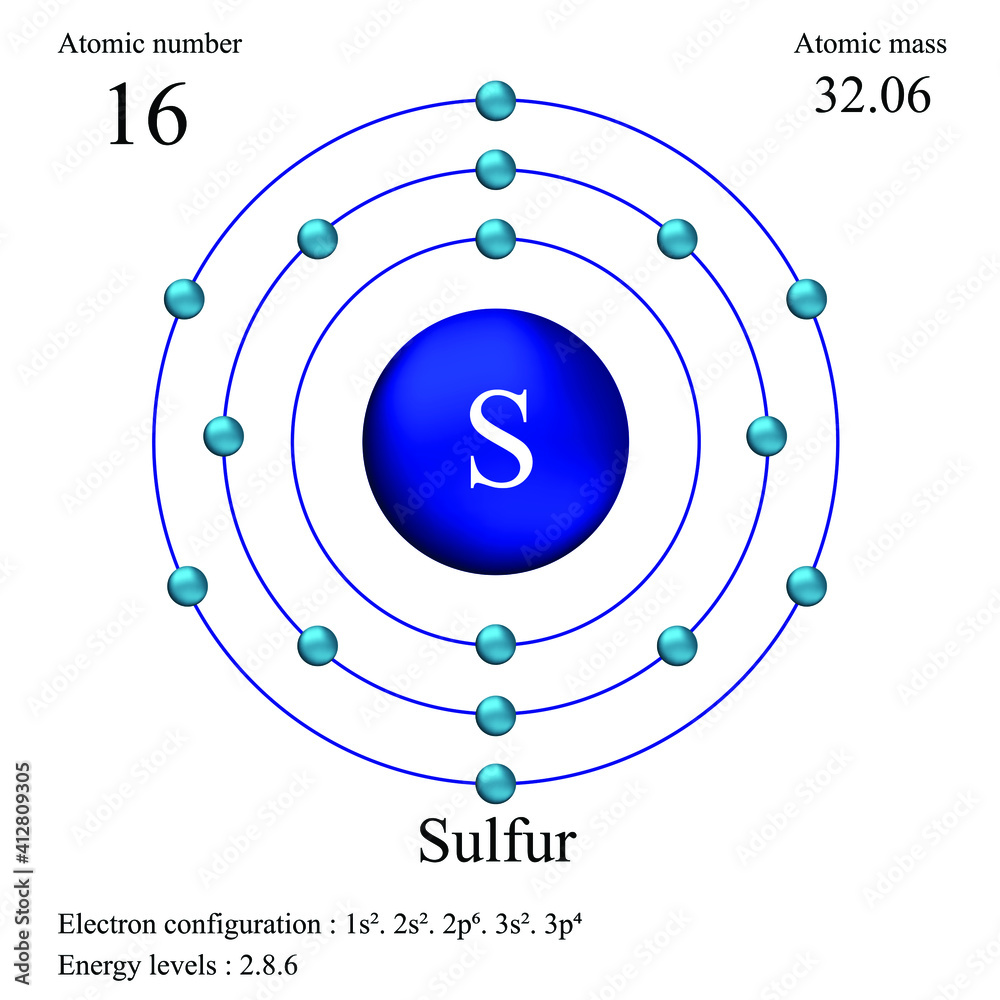
Sulfur atomic structure has atomic number, atomic mass, electron configuration and energy levels. Stock Vector | Adobe Stock
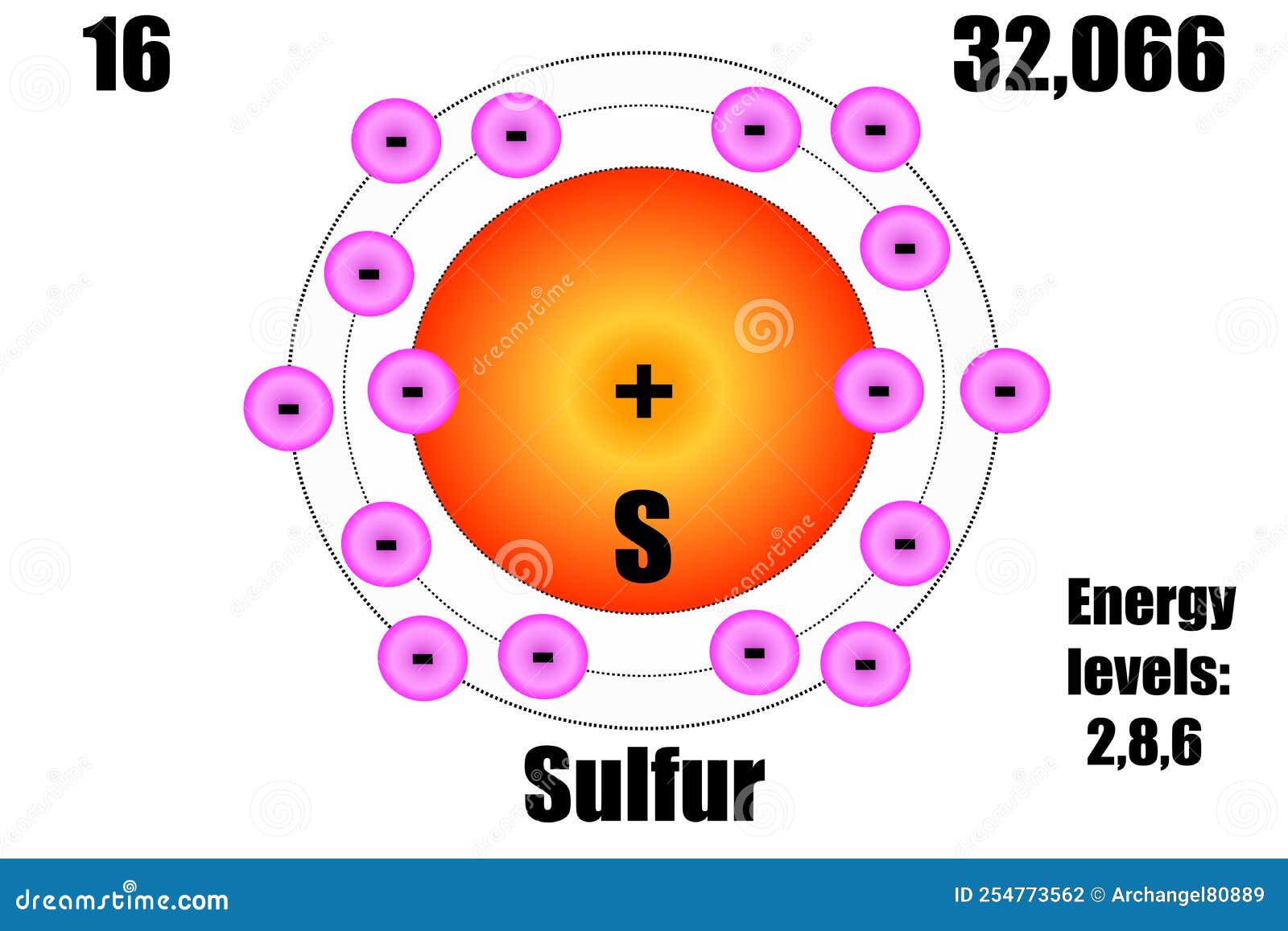
Sulfur Atom, with Mass and Energy Levels. Stock Vector - Illustration of formula, chemical: 254773562

PPT - 1. How many total electrons are in a neutral atom of Sulfur ? PowerPoint Presentation - ID:2465357